
QCM 2ème partie. N de QUESTIONNAIRE : 006. Examen du 21 Avril Code réservé au CIFMD : - PDF Free Download

PDF) E-pharmacovigilance: development and implementation of a computable knowledge base to identify adverse drug reactions | Kay Brune - Academia.edu

Questionnaire formation ADR transport matières dangereuses : qcm examen théorique Adr - 90km | Actualité - transport - formation - examen
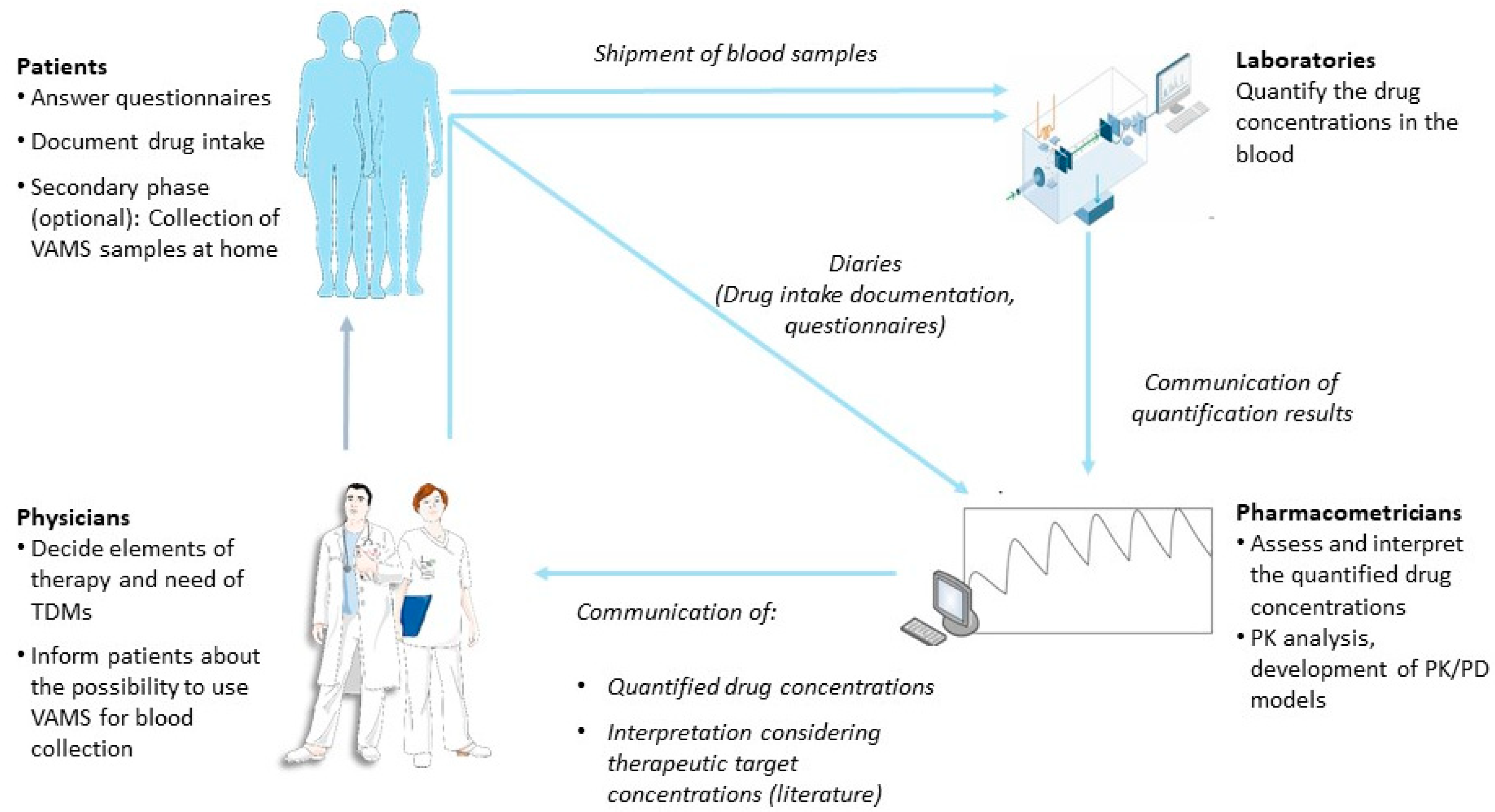
Cancers | Free Full-Text | Developing a Nationwide Infrastructure for Therapeutic Drug Monitoring of Targeted Oral Anticancer Drugs: The ON-TARGET Study Protocol

Are you susceptible to adverse drug reactions? | by Chiao-Feng Lin | DNAnexus Science Frontiers | Medium

PDF) Awareness related to reporting of adverse drug reactions among health caregivers: A cross-sectional questionnaire survey

E-pharmacovigilance: development and implementation of a computable knowledge base to identify adverse drug reactions. - Abstract - Europe PMC
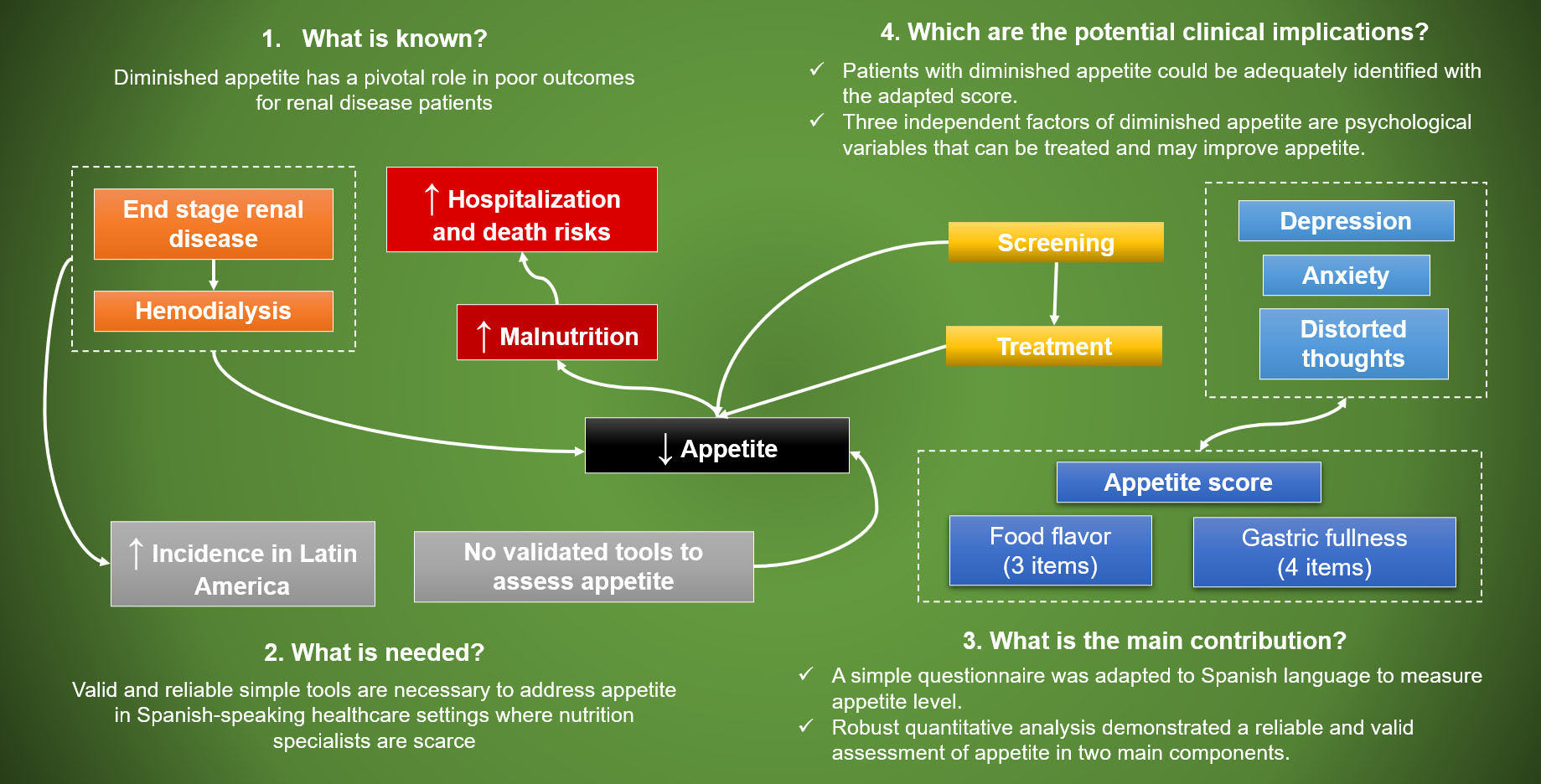
Nutrients | Free Full-Text | Assessment of Factors Related to Diminished Appetite in Hemodialysis Patients with a New Adapted and Validated Questionnaire

QCM 2ème partie. N de QUESTIONNAIRE : 006. Examen du 21 Avril Code réservé au CIFMD : - PDF Free Download

PharmActa: Personalized pharmaceutical care eHealth platform for patients and pharmacists - ScienceDirect

Development and validation of a questionnaire for the assessment of the knowledge, management and reporting ADR in paediatrics by healthcare teams (QUESA-P) | BMJ Open

An evaluation of knowledge, attitude and practice of Indian pharmacists towards adverse drug reaction reporting: A pilot study – topic of research paper in Clinical medicine. Download scholarly article PDF and read

Knowledge, attitude and practice among medical students related to Pharmacovigilence | World Journal of Pharmaceutical Sciences - Academia.edu

PDF) Development and Validation of Questionnaire to Assess The Knowledge, Attitude and Practice towards Adverse Drug Reactions Reporting among Healthcare Professionals




.jpg)


.jpg)